The Hidden Warning of Fall Colors
Did autumn reds and yellows evolve to repel insects?
By Brian Gallagher
Sign up for our monthly newsletter!
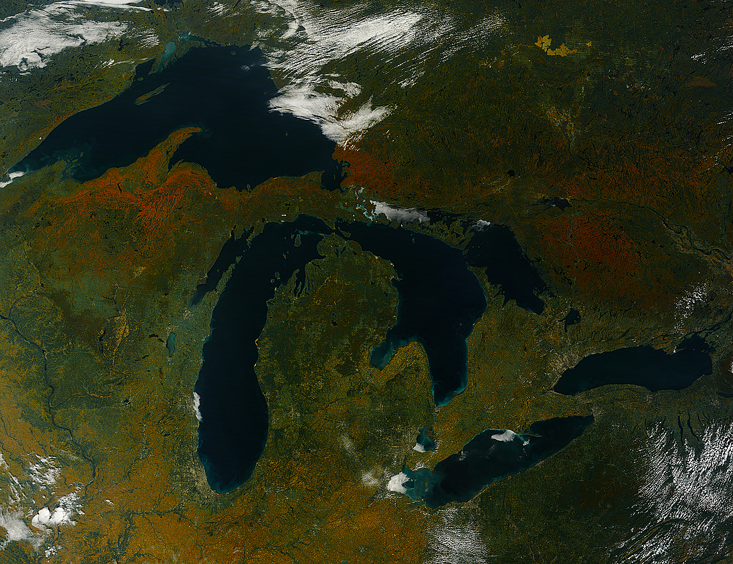
Drifting above North America last autumn, a NASA satellite named Terra partook in some high-altitude leaf peeping. In an aerial photograph snapped in September, swaths of orange and red saturate the green landscape, as if igniting the planet in a smokeless blaze. If an alien spacefarer were to happen upon this annual scene, it might mistake the spectacle for an intentional signal—a battle flare, perhaps, or a distress call. Earth-bound scientists, on the other hand, long dismissed autumn colors as a mere coincidence. “Such biochemical extravagancies … seem to have no function in the trees,” wrote one plant physiologist in 2000, echoing the sentiment of the 19th-century Scottish biologist Marion Newbigin.
But in the late 1990s, William D. Hamilton, an evolutionary theorist at Oxford, proposed that trees’ vibrant fall hues were indeed a signal, meant to ward off parasites that lay their eggs in autumn. The behavior, he reasoned, must be an outcome of coevolution, in which two or more species reciprocally drive one another’s adaptations. Plants and their pollinators are a classic case. “A flower and a bee might slowly become, either simultaneously or one after the other, modified and adapted to each other in the most perfect manner,” Charles Darwin wrote in The Origin of Species. A change in the flower causes a change in the bee, which causes a change in the flower … and so on, until at last, the process of pollination is born.
There must be a reason why some dying leaves evolved colorful fall displays while others didn’t.
Hamilton suggested that autumn colors evolved by a similar route. In tree species that suffer greatly from insect damage, he argued, natural variation would have produced some individual trees with stronger defenses—toxins perhaps, or bad tastes—as well as coincidentally brighter leaves. Some of these trees’ insect attackers would have evolved to read the trees’ colorful displays as warning signs and colonized duller, less-defended trees instead. In this scenario, the most colorful trees and the cleverest bugs would have been the most likely to survive, passing on their genes and their behavior.
Hamilton died in 2000, but his ideas, published the following year, ignited serious debate.1 “The field was initially very confrontational—botanists against ecologists, accusing each other of being idiots,” remembers Marco Archetti (now at the University of East Anglia, in England), who helped Hamilton model the signaling theory as an undergraduate. Now, after more than two decades of gathering evidence and counterevidence, advancing arguments and refuting them, experts still haven’t come to a consensus. Even Archetti admits that the theory doesn’t really hold up for yellow leaves. But red leaves might just fit Hamilton’s narrative. The color of blood and stop signs and poison dart frogs, red could be just as alarming in autumn leaves as it is most everywhere else.
Ecology textbooks often explain fall coloration as an incidental byproduct of leaf death. Deciduous trees shed their leaves in autumn to prepare for winter, when freezing temperatures and shorter days reduce the quantity of chemical energy they are able to manufacture from sunlight. Before its leaves fall, however, a tree—in a kind of self-cannibalism—will break down chlorophyll, a green photosynthesizing pigment, into nutrients such as nitrogen, which are absorbed into the trunk and stored for spring. Timing is crucial: If the tree kills off its leaves too early, it misses out on sunlight. If it waits too long, its green leaves will frost over, preventing it from absorbing their nutrients.
As trees harvest their own chlorophyll, other pigments in their leaves are unmasked or synthesized. Carotenoids, for example, absorb violet, blue, and green light, reflecting yellows and oranges. In spring and summer, they aid in photosynthesis, but because they are less concentrated than chlorophyll, their colors are hidden. In fall, trees absorb carotenoids more slowly, if at all, allowing their yellow and orange hues to shine through. Anthocyanins, another class of pigments in some fall leaves, give foliage a reddish, or sometimes purplish, hue. Unlike carotenoids, they’re not present during the growing season but instead are produced during the dying process.
Clearly, color change and leaf death are closely linked. But, Hamilton and Archetti reasoned, they don’t have to be. Maples and oaks exhibit brilliant autumn shades while other trees in the same climate, such as firs and spruces, are evergreen, continuously shedding leaves and growing new ones throughout the year. There must be a reason, the researchers presumed, why some dying leaves evolved colorful fall displays while others didn’t.
If trees wanted to deter herbivorous insects, yellow leaf coloration is about the worst strategy they could pick.
With Hamilton’s encouragement, Archetti set to work creating a model using game theory, according to which, he says, “your best decision depends on what other participants in the situation will decide.” Take, for instance, the male peacock’s tail feathers. Game theory shows that males evolved large, colorful tails because they attracted females, but only the fittest males could afford the flashiest displays because big tails made it harder to escape predators. Females, in turn, chose to mate with the most ostentatious males because the females evolved to trust this pageantry as a sign of genetic strength.
Archetti believed that game theory could similarly describe the evolutionary faceoff between trees and insects. Just as male peacocks must “decide” how many resources, if any, to invest in a large tail, trees must “decide” how many resources are worth investing in an autumn spectacle. Insects, meanwhile, must decide which trees are the most hospitable, just as female peacocks must decide which potential mates are the most fit. Archetti reasoned that in order for fall colors to evolve as a trustworthy signal, like the peacock’s tail, they must be useful as well as burdensome. Otherwise, trees would display them willy-nilly, and the message would lose its meaning.2

Validating Archetti’s model required empirical data. Before he died, Hamilton and Sam Brown, a zoologist at Oxford, managed to dig up supporting evidence from the scientific literature. In their 2001 paper, they described how the production of bright autumn colors could indeed be costly, as Archetti had predicted. One study showed that before dropping their leaves, Norway Maples absorbed no more than 50 percent of the leaves’ carotenoids. Hamilton and Brown surmised that the lost pigments could have gone to good use as an energy source, yet were forsaken as a warning signal instead, keeping the leaves yellow longer. Trees, they maintained, also lose precious photosynthesizing time by sacrificing chlorophyll early in autumn to unveil colorful signals that last for weeks or months. And trees that manufacture new color compounds, including anthocyanins, fluorescent chemicals, and optical brighteners, use up energy in the process.
Hamilton and Brown also named an insect family that they thought may have helped drive the evolution of fall colors in some trees. Aphids, a sap-sucking pest common to temperate regions worldwide, can spread viruses, stunt a tree’s growth, and reduce its bulk by as much as two thirds. The researchers argued that because aphids sometimes migrate and colonize new plants during autumn, precisely when leaves change color, they could have evolved to read this change as a signal. What’s more, some aphid species can distinguish between bright and dull hues, particularly among yellows, and have a preference for more diluted shades.
Many plant biologists, however, were skeptical.3 They pointed out that aphids migrate during seasons other than fall, suggesting that their choice of tree often had nothing to do with color. The critics suggested instead that trees deploy anthocyanins and carotenoids as sunscreens during the fall, when their leaves are most vulnerable to sun damage due to environmental conditions.
Some experts maintain that the evolution of fall colors, even red, has nothing to do with insects.
Then in 2007, Thomas Döring, an expert in insect vision at Humboldt University, in Berlin, and a colleague took Hamilton and Brown to task for their assumptions about aphid color vision. Laboratory experiments with green peach aphids had shown that the insects will “enthusiastically try to drill their proboscides into surfaces that reflect green to yellow light,” Döring and his co-author wrote in the journal PLoS ONE.4 If yellow was a signal to stay away, these bugs weren’t getting the message. In his own research, Döring discovered that yellow light stimulates the same photoreceptors in aphids as green light, implying that the bugs probably can’t tell much difference between green and yellow leaves. “If trees wanted to deter herbivorous insects using color, yellow leaf coloration is about the worst strategy they could pick,” the authors concluded.
Archetti wasn’t convinced. He persuaded Döring to collaborate on an experiment to test which colors aphids actually preferred, based on their behavior in the field.5 On a patch of bare, sandy soil, the researchers set up 140 water dishes to trap aphids, each painted one of 70 autumn colors. When the results came in, Archetti had to concede he’d been wrong about yellow: The yellow dishes attracted even more aphids than the green ones. To aphids, yellow seemed to be just a “super-bright green,” Archetti explains. Carotenoids were knocked out of the coevolutionary theory game. The theory, however, still held up for red anthocyanins: The insects avoided the red-hued traps.

The ideal test of Hamilton and Archetti’s theory would be a controlled trial: Take two groups of red-leaf trees, expose only one group to insect pests, and observe them for many generations. If red is a warning signal, then you would expect this trait to disappear in the population of trees freed from the selective pressure of insect predation. Of course, the experiment would be impractical—no scientist today would live to see its end. Archetti, however, found a clever workaround.
In a 2009 paper, he compared populations of wild apple trees with their domesticated relatives, which farmers began breeding 2,000 years ago.6 Insects still attack domestic trees, but humans control tree breeding. Whether or not a tree avoids an insect infestation therefore has much less bearing on whether it produces progeny. Archetti reasoned that, without this adaptive pressure, red leaves would eventually be bred out of the apple tree. Indeed, this evolutionary reversal already appears to be happening: In Central Asia, Archetti reported, leaves turn red in 62 percent of wild apple trees but in only 39 percent of domesticated ones. Likewise, he points out, wild apricot and walnut trees in Asia tend to turn red-orange while European cultivations remain green or turn yellow.
According to Archetti’s game theoretic model, the red-hued trees should also be the most harmful to insects. Archetti tested this hypothesis by observing aphid colonies on 40 apple trees with green leaves, 40 with yellow leaves, and 40 with red leaves. The bugs settled the trees in fall, and by summer, about two-thirds of the offspring from the green-leaf colonies, and about half from the yellow-leaf colonies, survived. In red-leaf colonies, however, the survival rate was only one-third, suggesting that some unknown feature of red-leaf trees was interfering with breeding. In 2012, researchers in New Zealand studying the light brown apple moth, another apple tree pest, showed that a diet of red leaves killed moth larvae or significantly slowed their development.7
Still, some experts maintain that the evolution of fall colors, even red, has nothing to do with insects. “There’s a bias among evolutionary biologists to see color as a visual signal,” when often it is just a side effect of cellular processes, says David Lee, a retired botanist, previously at Florida International University. Other experts no longer see a point in arguing. “Actually there is not a conflict if autumn colors have more than one primary function,” says Jarmo Holopainen, an ecologist at the University of Eastern Finland. Kevin Gould, an ecophysiologist in New Zealand, agrees. Protection from sun damage may have been “the primary driving force for the evolution of red leaves,” he says, “but the pigments may have been co-opted for other functions,” such as deterring pests.
Part of the lasting power of Hamilton’s hypothesis may be its aesthetic appeal. Even Marion Newbigin, who more than 100 years ago saw no evidence that autumn coloration was “of the slightest use,” couldn’t resist the possibility that Darwinian forces were at work. “And yet,” she wrote in 1898, “it often displays to an extraordinary degree that beauty and perfectness which we are accustomed to regard as the result of the action of Natural Selection.”
Brian Gallagher is an assistant editor at Nautilus.
This article previously appeared in Nautilus.
References
1. Hamilton, W.D. & Brown, S.P. Autumn tree colours as a handicap signal. Proceedings of the Royal Society B 268, 1489-1493 (2001). ⤶
2. Archetti, M. The origin of autumn colours by coevolution. Journal of Theoretical Biology 205, 625-639 (2000). ⤶
3. Schaefer, H.M. & Wilkinson, D.M. Red leaves, insects and coevolution: A red herring? Trends in Ecology and Evolution 19, 616-618 (2004). ⤶
4. Chittka, L. & Döring, T.F. Are autumn foliage colors red signals to aphids? PLoS One Biology 5, e187 (2007). ⤶
5. Döring, T.F., Archetti, M., & Hardie, J. Autumn leaves seen through herbivore eyes. Proceedings of the Royal Society B 276, 121-127 (2009). ⤶
6. Archetti, M. Evidence form the domestication of apple for the maintenance of autumn colours by coevolution. Proceedings of the Royal Society B 276, 2575-2580 (2009). ⤶
7. Markwick, N.P., et al. Red-foliaged apples affect the establishment, growth, and development of the light brown apple moth, Epiphyas postvittana. Entomologia Experimentalis at Applicata 146, 261-275 (2013). ⤶
Plantings

Embodying Heaven: Frankincense and Myrrh and Their Connection to Divinity
By Nuri McBride

A Voice for the Trees
By Mary Ellen Hannibal

The Real Cost of Planting Trees
By Lauren E. Oakes

Monet’s Garden in Giverny
By Gayil Nalls

Riveted by Nature’s Beauty
By Matsuo Bashō

Eat More Plants Recipes:
Roasted Cauliflower Snowflakes
By Ina Garten

As Ireland transitions from the rich, smoky scent of peat-burning to a more sustainable future, its olfactory heritage is evolving. What will become the next iconic aromatic symbol of Ireland?
Click to watch the documentary trailer.